Blog & News
New SHADAC Brief Looks at Changes in Federal Surveys During COVID Pandemic
August 27, 2021: The impacts of the coronavirus pandemic have been far ranging—on education, work, healthcare, and almost every other aspect of daily life. However, another effect of COVID that is just now coming to light is the interruption of data collection processes and falling response rates for yearly federal surveys that, among other measures, provide estimates of health insurance coverage for the United States population. Such surveys would, under normal circumstances, be ideally suited to measure changes to insurance coverage during such a tumultuous time.
The impacts of the coronavirus pandemic have been far ranging—on education, work, healthcare, and almost every other aspect of daily life. However, another effect of COVID that is just now coming to light is the interruption of data collection processes and falling response rates for yearly federal surveys that, among other measures, provide estimates of health insurance coverage for the United States population. Such surveys would, under normal circumstances, be ideally suited to measure changes to insurance coverage during such a tumultuous time.
In a new brief, SHADAC summarizes COVID-era changes and challenges for four major federal surveys—the American Community Survey (ACS), the Current Population Survey (CPS), the Medical Expenditure Panel Survey (MEPS), and the National Health Interview Survey (NHIS)—such as shifting in-person collection methods to phone and email, adding COVID-related questions to questionnaires, extending survey fielding in order to address falling response rates, and delayed data release dates, among others.
All four surveys made significant changes to their Survey Operations:
In-person: The ACS, CPS, and NHIS all suspended in-person operations from March to June 2020, with limited in-person interviews resuming in either July or September 2020. The MEPS Insurance Component (MEPS-IC) suspended in-person interviews entirely.
Mailing: Mailing centers for the ACS and the CPS were shut down in March 2020, but re-opened with limited staffing in July 2020, allowing previously completed surveys to be collected and counted. The MEPS Household Component (MEPS-HC) shifted to dual web-and-mailing collection methods in the fall of 2020 while the MEPS-IC shifted to dual web-and-phone data collection at that time.
Telephone: All surveys (ACS, CPS, MEPS-HC and MEPS-IC, and NHIS) shifted to phone-only surveys while in-person fielding was shut down, or while mailing operations were suspended, in the case of the MEPS-IC.
Web: While both the MEPS-HC and MEPS-IC took a different direction and shifted to telephone-only survey collection in March 2020, the former proposed a shift to dual web-and-mailing data collection modes while the latter opted to shift to dual web-and-phone methods in the fall of 2020.
Three surveys made changes to their Questionnaires:
The ACS and the MEPS-IC opted not make any changes to their questionnaires in response to COVID-19.
The CPS added five new questions regarding COVID’s impact on employment to the questionnaire. The NHIS similarly added new questions to its survey form, but instead asked respondents about COVID testing, contraction and symptoms, and prevention measures.
The MEPS-HC was the only survey to create an entirely new questionnaire: the “Social and Health Experiences” survey, which asked respondents about changes or delays in care due to COVID.
Each survey experienced falling Response Rates:
Where they were able to be measured, survey response rates dropped significantly. Neither the MEPS-HC nor the NHIS has yet released response rates for 2020, but previous years show a drop in response rates for the latter survey of 20 percentage points (80 percent in 2018 to 60 percent in 2019). Both the ACS and CPS saw falling response rates between 2019 and 2020. The ACS dropped 15 percentage points (86 percent in 2019 to 71 percent in 2020), while the CPS fell by roughly 6 percentage points (67.6 percent in 2019 to 61.1 percent in 2020). The MEPS-IC saw a smaller decrease between the years, from 59.6 percent in 2019 to 56.1 percent in 2020.
SHADAC plans to continue monitoring reported changes to these and other federal surveys in 2020, and continuing into 2021 where applicable. As data from 2020 becomes available, our researchers will continue to produce resources to explain what effects these adaptations may have for understanding the data, and if alternative sources may provide ways to fill in gaps where 2020 data may be incomplete or unable to be reliably used.
Blog & News
Drug overdose deaths grew by almost 30 percent in 2020
July 15, 2021:Fentanyl- and methamphetamine-type drugs surged roughly 50 percent in 2020
Drug overdose deaths surged in the United States during the first year of the COVID-19 pandemic, increasing nearly 30 percent in just 12 months. Provisional data recently published by the U.S. Centers for Disease Control and Prevention (CDC) show that more than 92,000 people died of drug overdoses in 2020—surpassing records yet again.1
The growth was widespread throughout the country, with only two states (New Hampshire and South Dakota) spared from the jump in drug overdose deaths. Conversely, some states saw their death rates increase more than 50 percent, including Kentucky, South Carolina, Vermont, and West Virginia.
Much of the growth in drug overdose deaths was driven by synthetic opioids such as fentanyl, which increased more than 50 percent from 2019 to 2020 (see Figure 1). Fentanyl has become a key product for international drug traffickers, often finding its way as an adulterant in other drugs like heroin and cocaine, and even as an ingredient in counterfeits of common opioid prescription pills such as Oxycontin. The emergence of fentanyl in the U.S. illicit drug trade is a newer phenomenon beginning in the past decade, and it has recently spread from eastern states to increasingly affect states in the western half of the country as well.
Figure 1. Changes in drug overdose deaths in the U.S., 2019 to 2020
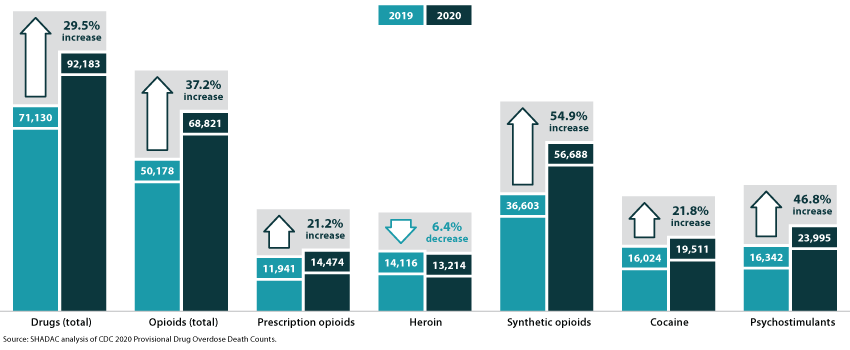
A family of drugs called “psychostimulants”—mostly methamphetamine—also drove a large increase in deaths in 2020, up nearly 50 percent since 2019. Deaths involving methamphetamine and other psychostimulants have grown dramatically in the past few years. The increased death toll involving psychostimulants is likely caused by two factors: First, the methamphetamine trafficked in the U.S. today is generally much more potent than methamphetamine sold in the past, raising the potential risk of overdoses caused by methamphetamine. Second, methamphetamine today is often contaminated with, or used alongside, synthetic opioids, raising the risk of an overdose involving the use of multiple drugs simultaneously.
Of the main drugs involved in overdoses2, only heroin was associated with a decline in deaths during 2020—falling by less than 10 percent since 2019. Meanwhile, overdose deaths involving prescription opioids increased more than 20 percent, reversing a trend of relatively stable or even declining death rates over several years. Cocaine overdose deaths similarly increased by more than 20 percent in 2020.
1 National Center for Health Statistics (NCHS). (2021, July 14). Vital Statistics Rapid Release: Provisional Drug Overdose Death Counts [Data set]. Centers for Disease Control and Prevention (CDC). https://www.cdc.gov/nchs/nvss/vsrr/drug-overdose-data.htm
2 The drug overdose death categories presented in the CDC data include: heroin, natural opioid analgesics (e.g., morphine and codeine) and semisynthetic opioids (e.g., oxycodone and hydrocodone), synthetic opioids such as methadone and synthetic opioids other than methadone (e.g., fentanyl and tramadol), cocaine, and psychostimulants.
Pages
- « first
- ‹ previous
- 1
- 2
- 3